Integrated Production – Our Greatest Strength
The highly integrated material loops at its major sites in Burghausen, Nünchritz and Zhangjiagang are one of WACKER’s key competitive advantages. Basically, integrated production involves using the byproducts from one stage as starting materials for making other products. The auxiliaries required for this, such as silanes, are recycled in a closed loop. Waste heat from one process is utilized in other chemical processes. Integrated production not only cuts energy and resource consumption, but also improves the use of raw materials in the long term, and integrates environmental protection into our processes.
Our integrated production system is primarily based on salt, silicon, methanol, acetic acid and ethylene as starting materials. In our integrated processes, we optimize material efficiency by purifying byproducts and reusing them or making them available for external use. Examples:
- In our integrated ethylene production system, we use ethylene to obtain organic intermediates, which we then turn into polymer dispersions and dispersible polymer powders.
- Our integrated silicon production system operates along similar lines. Although comprising only a small number of raw materials – silicon, methanol and salt (sodium chloride) – this system enables us to manufacture over 2,800 different silicone products, as well as pyrogenic silica and polysilicon.
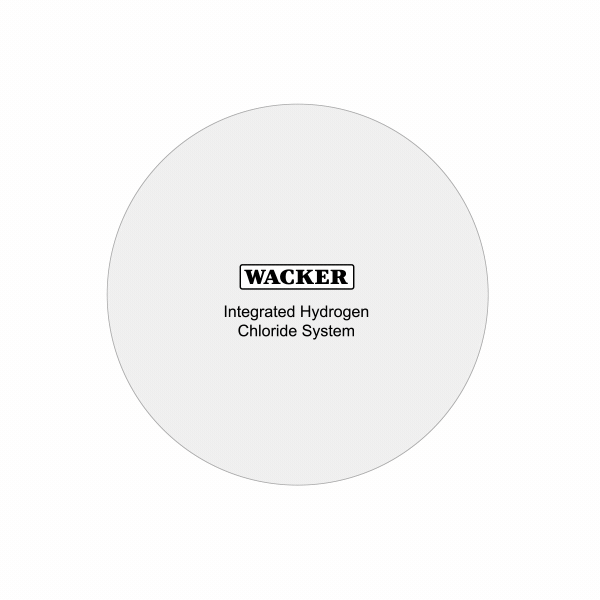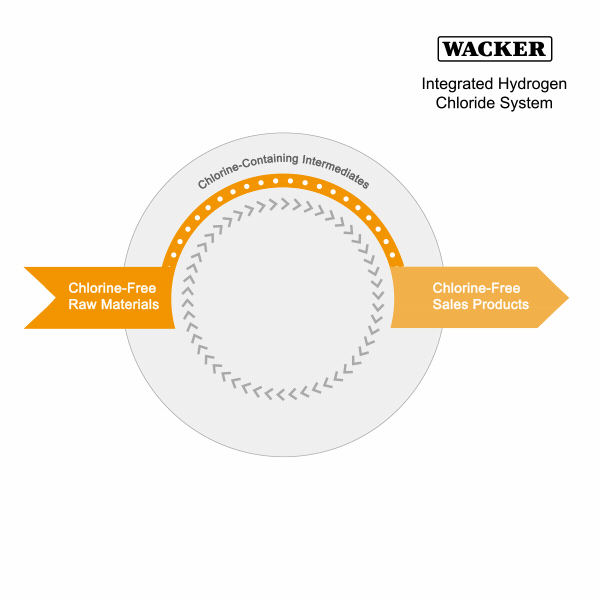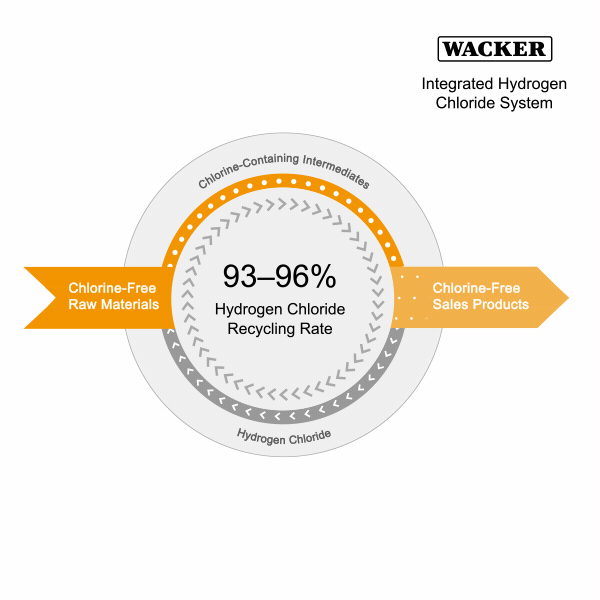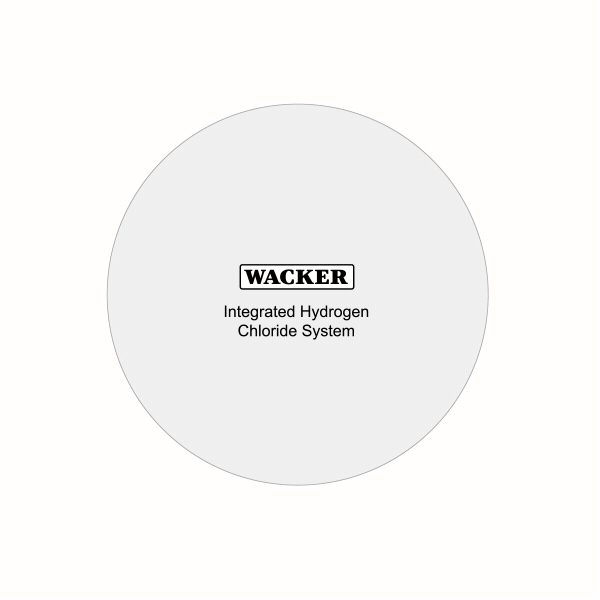
Integrated Hydrogen Chloride System
A further focus of our integrated production is to minimize hydrogen chloride consumption. Hydrogen chloride is an essential auxiliary deployed in the production of reactive intermediates from energy-poor natural materials. We then use these intermediates to make our end products.
Hydrogen chloride production requires a lot of energy. In our integrated material loop, we recover both hydrogen chloride and some of the energy in the form of heating steam during the conversion of the chlorine-containing intermediates to chlorine-free end products (such as silicones, hyperpure silicon or pyrogenic silica). We then return the recovered hydrogen chloride to the production loop and reuse it. This closed material loop reduces emissions and, due to lower raw-material consumption, shipment journeys.
We use a chlor-alkali membrane process to supply chlorine, hydrogen, caustic soda and hydrogen chloride as starting materials to our Burghausen site. This membrane electrolysis has enabled us to stop using mercury-based chlorine electrolysis and simultaneously cut energy consumption by around 25 percent per year. Thus, WACKER has fulfilled the chemical industry’s voluntary commitment to phase out mercury-based processes by 2020 at the latest well ahead of schedule. In 2017, we increased energy efficiency by a further 8 to 10 percent by using new electrolysis cells.
Examples of savings potential for resources through our integrated production system:
- We recycle 93 to 96 percent of the hydrogen chloride that we use in the production loops at our Burghausen and Nünchritz sites. During the reporting period, we again worked on closing our material loops as much as possible and reducing the loss rate by means of optimization projects.
- In 2018, our integrated hydrogen chloride system in Burghausen prevented the emission of over 495 kilotons (kt) of CO2 equivalents (CO2e) (2017: 719 kt). The CO2e effect dropped from 2017 to 2018, because we equipped our chlor-alkali electrolysis with more efficient membranes. These require less energy specifically to generate chlorine and, from it, hydrogen chloride. Due to the high reutilization rate, we save on fresh hydrogen chloride and, consequently, on raw materials and energy.
- We optimized the hydrogen loops in our integrated polysilicon production system and thus significantly lowered the consumption of hydrogen extracted from natural gas. This has led to a reduction in carbon dioxide emissions of 8,000 metric tons per year compared with 2016.
Zhangjiagang in China – alongside Burghausen (Bavaria) and Nünchritz (Saxony) in Germany – is our third major integrated production site. We employ state-of-the-art environmental technology in China, too, where we operate facilities according to stringent national and WACKER EH&S standards.
This also applies to our new site in Charleston in the USA, where we are further optimizing our system of integrated polysilicon production. We have erected a new production plant for HDK® pyrogenic silica there. This constitutes an important addition to the supply chain at the Charleston site, because the main byproduct of polysilicon manufacturing is tetrachlorosilane, which we can either convert and feed back into the production loop or we can use it to create added value by processing it further into HDK®. As already operated by WACKER at its Burghausen and Nünchritz sites in Germany, an integrated production system that combines polysilicon and HDK® facilities permits maximum flexibility in the processing of tetrachlorosilane, avoids the disposal of waste products and thereby enhances the viability of integrated production as a whole.