Products
New Goal
90% of products with neutral or positive sustainability contribution in 2030 (in line with WACKER Sustainable Solutions)
Corporate entity: Groupwide
Deadline: 2030
Outlook
Product Safety
During the next reporting period, we will continue to focus on the following:
- Promptly registering materials subject to REACH and updating the registration dossier
- Implementing GPS (the Global Product Strategy of the International Council of Chemical Associations, or ICCA) through publication of GPS Safety Summaries for all relevant substances registered under REACH
Another regulatory focal point is chemical nanomaterials. The number of national registers in which nanomaterials are recorded is increasing and will further augment the administrative workload for companies active in this market.
The United Kingdom’s departure from the European Union has rendered previous REACH registrations in this region no longer valid. For this reason, the UK has developed and enacted its own system for regulating chemicals – one that largely corresponds to EU regulations. All chemical substances contained in chemical products must now be registered separately in this new system.
Products
We are working toward establishing our strategy for the circular economy in the Group, focusing on three cornerstones:
- Renewable raw-material sources
- Production cycles
- Material cycles
In 2021, WACKER POLYMERS and WACKER SILICONES plan to update the life cycle assessments of their products.
With our SustainaBalance® sustainability strategy, we are aiming for climate neutrality by 2050.
Research & Development
The Group’s research and development work remains focused on key strategic projects. WACKER intends to spend 11 percent of its R&D budget on key projects in 2021 (2020: 10 percent). Our R&D work is prioritizing the highly promising fields of energy, electronics, automotive engineering, medical technology, consumer care, biotechnology and construction applications. We are devoting particular attention to efficient energy utilization, energy storage and renewable-energy generation.
WACKER BIOSOLUTIONS is working on adding human milk oligosaccharides (HMOs) for baby food and other applications to its product portfolio. Corporate R&D is developing processes for bioengineering HMOs.
In the first quarter of 2021, WACKER acquired pharmaceutical contract manufacturer Genopis Inc. from Helixmith Co Ltd. and Medivate Partners LLC (both in Seoul, South Korea) and renamed it Wacker Biotech US Inc. The company operates a fermentation line in San Diego, USA, and has extensive experience in the production of plasmid DNA (pDNA) according to GMP (Good Manufacturing Practice) quality guidelines. WACKER and Helixmith plan to collaborate on the production of a pDNA gene therapy for the treatment of diabetic peripheral neuropathy (DPN). The formulation – VM202 – was developed by Helixmith and is currently in clinical trials.
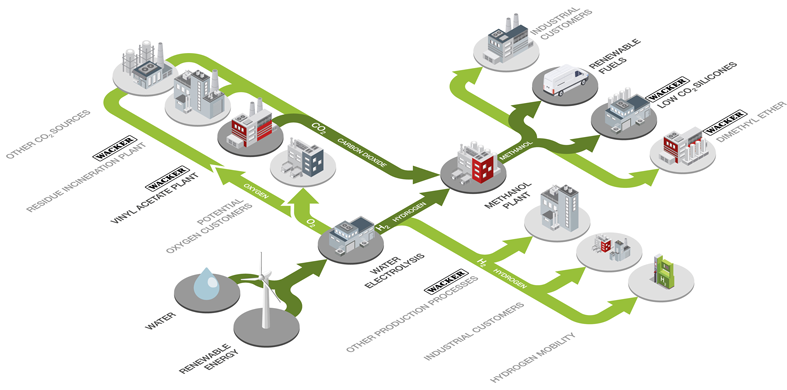
RHYME Bavaria
Wacker Chemie AG has applied to the European Commission for funding to produce green hydrogen and renewable methanol at its Burghausen site. The RHYME Bavaria (Renewable HYdrogen and MEthanol) project sees WACKER building a 20-megawatt electrolysis plant to produce hydrogen from water using renewable electricity. The project also includes a synthesis plant for processing hydrogen into renewable methanol, using carbon dioxide from existing production processes. Hydrogen and methanol are both key starting materials, for example for silicones. Compared with existing production methods using fossil-based methanol, the new processes could significantly cut CO2 emissions.